The definitive guide to ketogenic diet science: how your body transitions to ketosis
A complete analysis of the biochemical mechanisms that drive nutritional ketosis — from glycogen depletion and beta-oxidation to clinical applications and metabolic adaptation.
What is ketogenic diet science?
The ketogenic diet (KD) is a metabolic strategy characterized by a very low intake of carbohydrates, moderate protein consumption, and high fat intake. While recently popularized for rapid weight loss, the historical ketogenic diet science originated nearly a century ago as a therapeutic intervention for specific neurological conditions.
Understanding the complex biochemistry behind how the body shifts its primary energy source — a state known as ketosis — is essential for safe and effective implementation. The fundamental principle driving the KD is the drastic reduction of carbohydrate intake, typically limiting consumption to less than 50 grams per day, and sometimes even less than 20–30 grams depending on the individual.
When the body is deprived of its typical fuel source (glucose), it is forced into a metabolic state where it begins breaking down stored fat into compounds called ketone bodies, which then serve as an alternative energy supply for the body and brain. This profound shift makes the ketogenic diet a powerful metabolic intervention.
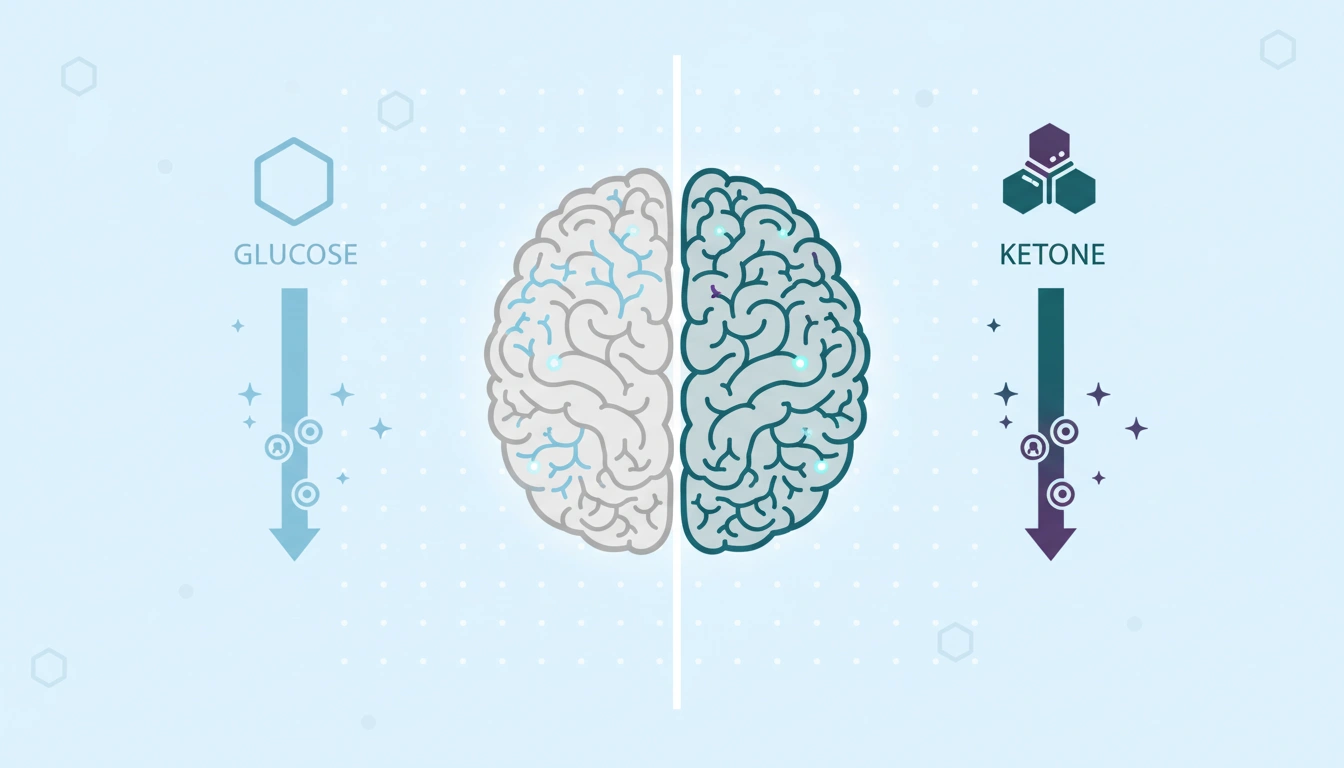
Understanding the metabolic shift: glucose vs. ketone metabolism
To appreciate the ketogenic diet science, one must first review basic cellular energy production. The human body generates energy in the form of adenosine triphosphate (ATP) through cellular respiration, which primarily relies on glucose. This process involves three main stages: glycolysis, the tricarboxylic acid (TCA) cycle (Krebs cycle), and the electron transport chain.
Carbohydrates: the body’s default fuel source
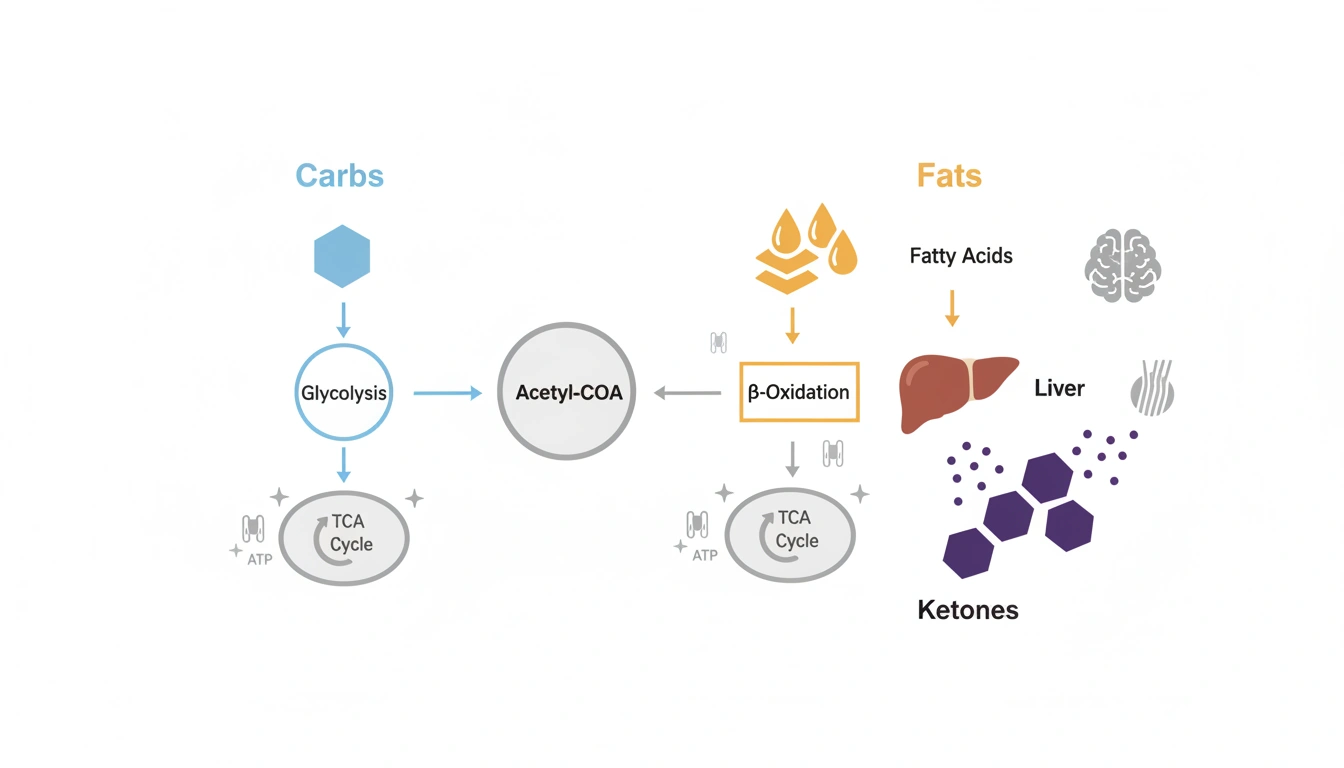
Under typical dietary conditions, carbohydrates are consumed and broken down into glucose — the primary and preferred metabolic substrate for most cells, including the liver and brain. Glucose molecules undergo glycolysis in the cell’s cytoplasm, producing pyruvate, which then enters the mitochondria to be converted into acetyl coenzyme A (acetyl-CoA). Acetyl-CoA subsequently enters the TCA cycle, culminating in the electron transport chain, where the bulk of ATP energy is generated.
Gluconeogenesis and glycogen depletion
When carbohydrate intake is severely restricted, the body’s easily accessible carbohydrate reserves — stored as glycogen in the liver — are rapidly depleted. Once these stores are exhausted, the body must produce its own glucose internally through gluconeogenesis. This process occurs mainly in the liver, using non-carbohydrate substrates such as pyruvate, lactic acid, glycerol, and specific amino acids.
However, endogenous glucose production is limited: proteins can only be converted to glucose by about 50%, and fats by only about 10% (due to their minimal glycerol content). This inability to rely heavily on fat for systemic glucose production means the body must find an alternative fuel source — and that is exactly what ketones provide.
Beta-oxidation: unleashing fat stores
When carbohydrates are scarce, the body enters a catabolic state, leading to the increased breakdown of fat — both dietary fat and stored triglycerides from adipose tissue. Triglycerides, composed of three fatty acids attached to a glycerol backbone, are broken down in the mitochondria by a process called beta-oxidation.
Beta-oxidation directly generates acetyl-CoA. Since the body is low on carbohydrates, this acetyl-CoA bypasses the initial stages of glucose metabolism and is routed toward an alternative pathway in the liver — ketogenesis — to produce usable fuel for the entire body, including the brain.

The core mechanism: ketogenesis and ketone bodies
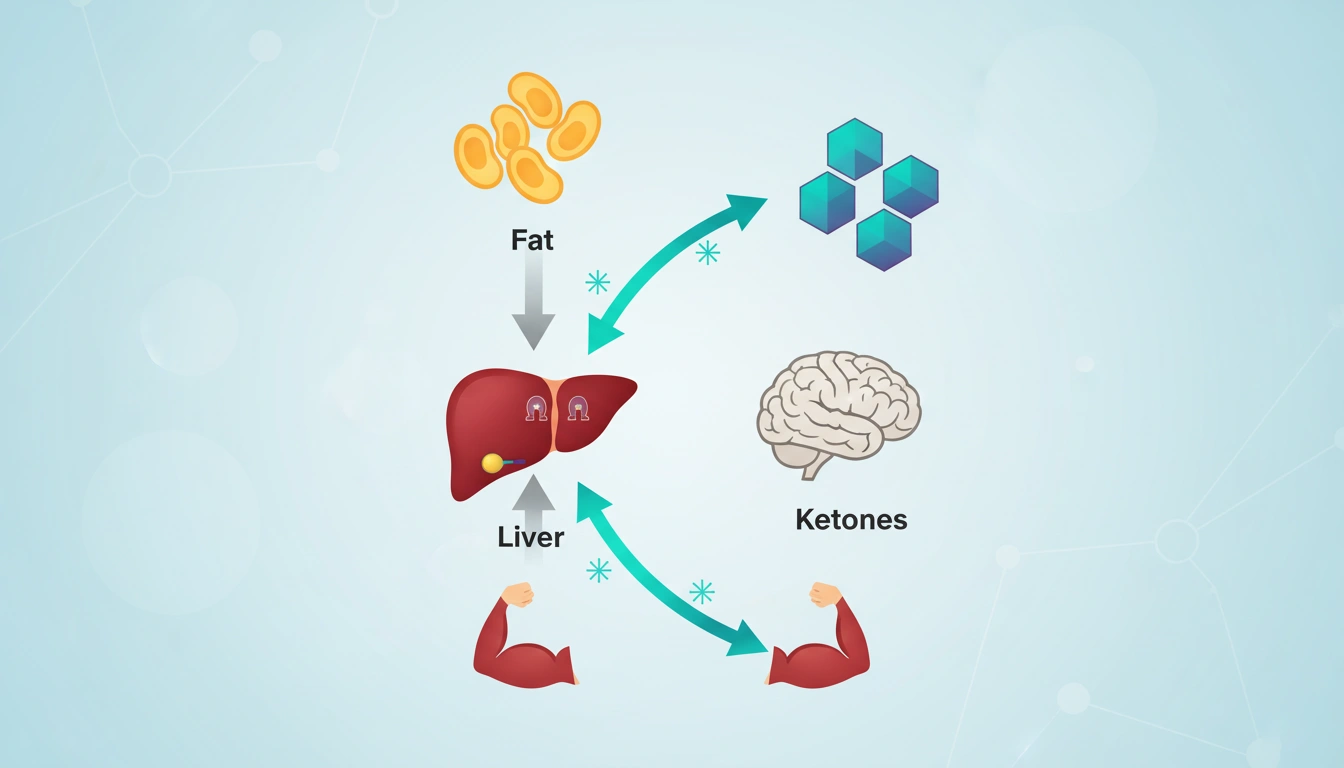
Ketosis is the metabolic state defined by an increased concentration of ketone bodies in the blood. This is the central tenet of ketogenic diet science. The liver is the primary site for the synthesis of ketone bodies — a process triggered by low insulin secretion and high levels of fatty acids resulting from limited carbohydrate intake.
The three principal ketone bodies
There are three primary ketone bodies produced during ketogenesis, each serving as a crucial replacement for glucose across different tissues:
The most abundant ketone body. A carboxylic acid commonly measured when tracking ketosis. The primary energy currency of nutritional ketosis.
Measured by urine strips. The precursor to both BHB and acetone. Elevated levels confirm active ketogenesis.
Detected in exhaled breath. Responsible for the characteristic “keto breath” reported by some dieters during early adaptation.
🔬 Why ketones are called “super fuel”
Ketone bodies, being water-soluble, can be transported through the bloodstream to vital organs — the heart, muscle tissue, kidneys, and brain can all effectively use them for energy. They are referred to as “super fuel” because they produce a greater amount of ATP compared to glucose: 100 grams of beta-hydroxybutyrate yields approximately 10,500 grams of ATP, while 100 grams of glucose produces only 8,700 grams. This mechanism allows the body to maintain efficient fuel production even in a caloric deficit.
Note: the liver itself cannot utilize ketones due to the absence of the necessary enzyme (diaphorase) — it produces them exclusively for export to other tissues.
Nutritional ketosis vs. ketoacidosis: a critical distinction
A common misconception associated with ketogenic diet science is the confusion between nutritional ketosis (a safe physiological state) and diabetic ketoacidosis (a life-threatening medical emergency). Understanding this distinction is fundamental to safe practice. See our full breakdown: Ketosis vs. Ketoacidosis.
| Parameter | Nutritional Ketosis ✅ | Diabetic Ketoacidosis ⚠️ |
|---|---|---|
| Cause | Controlled low-carb diet or fasting | Uncontrolled type 1 diabetes, prolonged alcoholism |
| Blood ketones | 0.5 – 5 mmol/L (max ~8 mmol/L) | 15 – 25 mmol/L or higher (>20 mmol/L) |
| Blood pH | Normal — body self-regulates | Drops significantly (acidosis) — medical emergency |
| Insulin | Small amount present; process is regulated | Near-zero insulin; uncontrolled ketone accumulation |
| Status | Safe / Physiological | Emergency / Pathological |
In a healthy individual, life-threatening ketoacidosis cannot happen as long as the person can generate even a small amount of insulin.
Physiological adaptation: the keto-adaptive state
The body’s shift to utilizing ketones is a gradual process — known as keto-adaptation — that takes at least a few weeks to complete.
Fueling the brain: ketones cross the blood-brain barrier
The brain is an energetically demanding organ, and while it normally prefers glucose, it can adapt to use ketone bodies — particularly beta-hydroxybutyrate — when glucose is scarce. Under sustained low-carbohydrate conditions, ketone bodies can represent up to 75% of the brain’s total energy supply.
The ability of ketones to cross the blood-brain barrier provides a reliable alternative energy source for neurological function. This neuroprotective mechanism is partially why ketogenic diet science has been historically applied to neurological disorders such as epilepsy, and is now being researched in Alzheimer’s and Parkinson’s disease.
Effects on appetite and satiety
One of the key mechanisms contributing to weight loss on the KD is its powerful effect on hunger and appetite regulation.
- Satiety hormones: the high fat and moderate protein content of the ketogenic diet promotes a greater feeling of satiety and fullness, which can lead to a spontaneous reduction in overall caloric intake — without conscious calorie counting.
- Ketone-induced anorexia: ketosis itself appears to suppress appetite. Research suggests that individuals adhering to the KD report less hunger and a reduced desire to eat, independent of caloric intake.
- Stable insulin levels: the steady, low insulin levels achieved on the diet help eliminate the sharp blood sugar fluctuations that typically trigger hunger cues in the brain on a high-carbohydrate diet.
New discoveries in ketone metabolism
Recent research has begun to uncover previously unrecognized complexity in how the body processes ketones. A collaborative study identified a metabolic “shunt pathway” where enzymes attach beta-hydroxybutyrate (BHB) to amino acids, producing a family of compounds called BHB-amino acids.
This discovery suggests that ketones have active roles beyond simple fuel. The BHB-amino acids were found to chemically resemble Lac-Phe — a molecule known to suppress appetite after strenuous exercise. Consequently, these BHB-amino acids suppress feeding behaviors and promote weight loss, indicating that this shunt pathway is crucial for the regulation of appetite and ketosis-associated weight loss. The identification of these metabolites provides a new framework for investigating the mechanisms behind the KD’s benefits, including seizure control.
Timeframe for keto-adaptation
The body’s shift from primarily burning glucose to efficiently utilizing fat and ketones is not instantaneous. Understanding the timeline helps set realistic expectations and reduce early dropout.
- Initial entry into ketosis: for most people, entering ketosis (detectable ketone levels) takes about 2 to 7 days of consuming fewer than 50 grams of carbohydrates daily.
- Full keto-adaptation: where the body and brain efficiently use ketones for sustained energy, typically takes at least a few weeks — often 4–8 weeks for complete metabolic recalibration.
- Factors influencing speed: age, metabolism, physical activity level, and previous diet all influence how quickly ketosis is achieved. Higher prior carb intake requires more time to fully deplete glycogen stores. Intermittent fasting can also help accelerate the process.
Clinical applications of ketogenic diet science
The ketogenic diet has validated therapeutic applications across multiple medical domains — from its 100-year history in epilepsy to emerging research in neurodegenerative disease.
Epilepsy treatment: the historical foundation
The original therapeutic application of ketogenic diet science was for the management of refractory (drug-resistant) epilepsy in children. Introduced in 1921 by Dr. Russell M. Wilder at the Mayo Clinic, the diet was designed to mimic the beneficial anti-convulsive effects observed during fasting.
Efficacy: the KD remains an accepted therapeutic option for pharmaco-resistant epilepsy, particularly in children. Studies show that approximately half of patients who try the diet achieve a seizure reduction of 50% or more, and about a third experience a reduction of over 90%. The effect can persist even after the diet is discontinued.
First-line indications: the KD is strongly considered for children who have failed to achieve seizure control with two or more anti-convulsant drugs. It is also a first-line therapy for specific metabolic diseases including pyruvate dehydrogenase (E1) deficiency and glucose transporter 1 deficiency syndrome (GLUT1-DS).
Benefits for type 2 diabetes and insulin sensitivity
Due to its drastic limitation of carbohydrates, the ketogenic diet is highly effective in controlling blood sugar and improving metabolic health parameters. Learn more in our keto and type 2 diabetes guide.
Improved glycemic control: studies demonstrate that the KD can significantly improve insulin sensitivity, with one older study reporting an increase of 75%. This improvement, coupled with weight loss, helps lower blood sugar and HbA1c (the marker of long-term glucose control).
Reduced medication use: in individuals with type 2 diabetes, the KD has been shown to help decrease or eliminate the need for diabetes medications — always requiring close medical supervision for safe adjustment.
Impact on cardiovascular risk factors
The effects of the KD on cardiovascular health are complex and sometimes controversial, but potential benefits have been reported in peer-reviewed literature. See also our guide on keto and cholesterol.
Lipid profile improvements (short-term): the KD can positively impact lipid profiles by reducing triglyceride levels and increasing HDL (“good”) cholesterol. Weight loss achieved on the diet may also help lower blood pressure.
LDL concerns: some studies indicate a potential initial increase in LDL cholesterol — particularly when the diet is high in saturated fats. This highlights the necessity of monitoring. Prioritizing healthy, unsaturated fats such as olive oil and fatty fish is recommended to mitigate this risk.
Neuroprotective potential in other conditions
Beyond epilepsy, the neuroprotective mechanisms of ketosis have prompted research into several other neurological and metabolic disorders.
Alzheimer’s and Parkinson’s disease: research suggests that ketones may help mitigate the neuroinflammation and oxidative stress often associated with neurodegenerative diseases. By providing an alternative fuel source, the KD may help palliate the hypometabolism suffered by neurons in these patients.
Other conditions under research: the KD is also being studied for potential therapeutic effects in polycystic ovarian syndrome (PCOS) — by reducing insulin levels and improving androgen markers — as well as traumatic brain injuries and certain types of cancer (though cancer research remains inconclusive).
Types of ketogenic diets: macronutrient science
Not all ketogenic diets are identical. Multiple protocols exist with different fat-to-protein-to-carbohydrate ratios, each suited to different goals and clinical contexts. See our full guide: types of keto diet: SKD, TKD, and CKD.
Standard and high-protein ketogenic diets
- Standard Ketogenic Diet (SKD): the most extensively researched version and the baseline for ketogenic diet science. Typical ratio: ~70% fat, 20% protein, 5–10% carbohydrates.
- High-Protein Ketogenic Diet: similar to SKD but with an increased protein ratio: often 60% fat, 35% protein, 5% carbohydrates.
- Classic Therapeutic KD (Epilepsy): follows a strict 4:1 ratio of fat weight to combined protein and carbohydrate weight — approximately 90% of calories from fat.
Modified and targeted approaches
- Modified Atkins Diet (MAD): does not restrict total calories or protein. Has a lower overall fat ratio (approximately 1:1). Restricts carbohydrates initially to 10–20 grams per day.
- Cyclical Ketogenic Diet (CKD): involves periods of high-carb refeeds — for example, 5 ketogenic days followed by 2 high-carb days. Used primarily by athletes.
- Targeted Ketogenic Diet (TKD): allows for the addition of carbohydrates specifically around workout times to support performance without exiting ketosis.
- MCT Diet: utilizes medium-chain triglyceride oils (often derived from coconut oil) because they are more ketogenic than standard long-chain triglycerides, allowing a slightly higher allowance of protein and carbohydrates.
The importance of protein moderation
Maintaining moderate protein intake is crucial for sustaining nutritional ketosis. Protein is often the most misunderstood macronutrient in keto. See our dedicated guide: how much protein on keto.
- Preventing gluconeogenesis: if protein consumption is too high (above roughly 35% of total calories, or 1.0–1.5 grams per pound of lean body mass), the body can convert excess amino acids into glucose. This rise in circulating glucose can interrupt the state of ketosis and reduce ketone levels.
- Kidney strain: excessive protein consumption forces the kidneys to excrete extra nitrogen, potentially straining individuals with pre-existing kidney conditions.
Dietary compliance: foods to embrace and avoid
Meals on the ketogenic diet should be based predominantly on whole, high-fat, and moderate-protein sources. For a deeper reference, see the top 10 essential keto foods and the complete keto vegetables list.
✅ Foods to prioritize (high fat, low carb)
- Healthy oils and fats: olive oil, coconut oil, avocado oil, butter, cream, and ghee.
- Fatty proteins: red meat, pork, ham, bacon, poultry, eggs (ideally pasture-raised or omega-3 enriched), and fatty fish — salmon, trout, mackerel, herring.
- High-fat dairy: unprocessed cheeses (cheddar, mozzarella, blue, cream cheese), sour cream, and full-fat Greek yogurt.
- Nuts and seeds: almonds, walnuts, flaxseeds, chia seeds, and pumpkin seeds.
- Low-carb vegetables: leafy greens (spinach, kale, arugula), cruciferous vegetables (broccoli, cauliflower, Brussels sprouts), asparagus, zucchini, bell peppers, tomatoes, and mushrooms.
- Low-carb fruits (in strict moderation): berries (strawberries, raspberries, blackberries) and avocado.
🚫 Foods to severely limit or eliminate (high carb)
- Sugars and sweets: soda, fruit juices, candy, ice cream, cake, and all sugary drinks or foods.
- Grains and starches: wheat-based products (bread, pasta), rice, oatmeal, cereals, and other grains.
- Starchy vegetables and tubers: potatoes, sweet potatoes, parsnips, carrots, and corn.
- Legumes: peas, beans, lentils, and chickpeas.
- Most fruits: all high-sugar fruits — bananas, apples, grapes, mango — are restricted.
- Processed foods: most diet or low-fat products, processed vegetable oils, and high-sugar condiments.
Risks and necessary medical monitoring
Navigating short-term side effects: the keto flu
As the body transitions into ketosis, individuals commonly experience the “keto flu” — a cluster of temporary symptoms that usually last from a few days up to two to four weeks. Common symptoms include:
- Fatigue and low energy: often attributed to the body’s struggle to adapt to fat as the primary fuel source.
- Headaches and brain fog: poor mental function and difficulty concentrating are frequently reported.
- Gastrointestinal distress: nausea, vomiting, diarrhea, and constipation are common during early adaptation.
- Electrolyte imbalance: the initial rapid weight loss is often water weight (due to glycogen depletion), which flushes out electrolytes — causing muscle cramps and dehydration.
Mitigation strategies
- Hydration and electrolytes: increasing water intake and replenishing electrolytes (sodium, potassium, magnesium) is crucial. Recommended intake during initiation: 3,000–4,000 mg sodium, 1,000 mg potassium, 300 mg magnesium daily. See our electrolytes guide.
- Gradual initiation: slowly lowering carbohydrate consumption over a few weeks helps the body adjust more smoothly and reduces symptom severity.
Long-term nutritional and organ health considerations
Long-term adherence to a KD, especially when unsupervised, raises significant concerns that should be discussed with a healthcare provider before starting:
- Nutrient deficiencies: because the diet severely restricts whole grains, legumes, and many fruits and vegetables, there is a risk of deficiencies in vitamin D, calcium, B vitamins (thiamine/B1, folate), iron, and magnesium. Supplementation is typically necessary.
- Kidney health and stones: the KD increases the risk of kidney stones due to acidosis (which increases calcium excretion), hypocitraturia, and low urine pH. People with pre-existing kidney insufficiency should not follow a high-protein diet as it can exacerbate their condition.
- Cardiovascular risk: sustained high intake of saturated fats and animal products is associated with increased cardiovascular risk in some populations and may worsen lipid profiles by raising LDL cholesterol.
- Liver function: people with pre-existing liver conditions may metabolize excess fat poorly. Hepatic steatosis (fat accumulation in the liver) is a reported long-term adverse effect in some cases.
- Potential accelerated aging (controversial): a mouse study suggested that long-term, sustained KD might accelerate aging of vital organs via cellular senescence and p53 pathway activation. However, this effect was reversible upon returning to a standard diet, and an intermittent KD schedule (4 days keto, 7 days standard) did not increase p53 activation.
Contraindications and the need for professional supervision
The ketogenic diet is not universally suitable. Consultation with a physician or specialized registered dietitian is mandatory before initiation, especially for those with underlying health conditions.
⚠️ The KD is strictly contraindicated for individuals with:
- Pancreatitis or liver failure/cirrhosis
- Disorders of fatty acid oxidation or fat metabolism (e.g., carnitine deficiencies, porphyrias, pyruvate kinase deficiency)
- Uncontrolled metabolic or homeostatic disorders
- Individuals taking SGLT2 inhibitors for type 2 diabetes (increased DKA risk)
- Children — except for specific medical indications like refractory epilepsy or metabolic disorders
- Pregnancy and lactation
Ketogenic diet science and weight management efficacy
Weight reduction on a KD is driven by multiple complementary mechanisms:
- Increased fat burning (ketosis): by drastically limiting glucose, the body is forced to increase the metabolism of stored fat for energy.
- Diuretic effect: the initial rapid weight loss (often 2–3 kg in the first few days) is largely water weight lost as stored glycogen reserves are depleted.
- Appetite suppression: the high fat and protein content, combined with the presence of ketones, reduces hunger and leads to a spontaneous reduction in caloric intake — the most critical factor for long-term weight loss.
- Metabolic expenditure: the body expends a significant amount of energy converting fats and proteins into necessary glucose (gluconeogenesis), contributing to additional caloric expenditure.
Short-term vs. long-term results
Studies consistently show that the KD leads to faster initial weight loss compared to traditional low-fat or Mediterranean diets. A meta-analysis comparing very low-carbohydrate KD to low-fat diets found superior weight reduction with KD in the long term. However, this advantage often diminishes over extended periods.
Many experts caution that the highly restrictive nature of the KD makes long-term adherence difficult, leading to potential “yo-yo dieting” — which is associated with increased health risks. A Mediterranean-style diet mixed with intermittent fasting is often advised for greatest longevity and lowest cardiovascular risk.
Implementation and monitoring: ensuring safety
Required professional team
Safe implementation of the KD involves an interprofessional healthcare team:
- Physician or neurologist: to screen for contraindications, monitor underlying conditions (especially heart and kidney health), and adjust medications — critical for diabetes patients.
- Registered Dietitian (RD): to calculate precise macronutrient ratios (ketogenic ratio, calorie needs, fluid intake), customize meal plans, and monitor for nutritional deficiencies. Use a free keto macros calculator as a starting point.
- Pharmacist: to advise on the carbohydrate content of necessary medications and supplements — many contain hidden carbs that can disrupt ketosis.
Monitoring ketosis
The physiological state of ketosis can be monitored to ensure the diet is effectively restricting carbohydrates and converting fat into fuel. For a full comparison of testing methods, see: measuring ketosis: blood vs. urine vs. breath.
- Testing methods: ketones can be measured in the blood (most accurately, focusing on beta-hydroxybutyrate), urine (using nitroprusside strips for acetoacetate), or breath (for acetone excretion).
- Optimal blood ketone range: blood ketone levels between 1.5 and 3.0 mmol/L are often considered the optimal ketosis range for desired therapeutic effects.
- Regular clinical labs: regular analysis of lipid status (total cholesterol, HDL, LDL, triglycerides) and monitoring of renal and hepatic function is crucial for long-term safety.
Conclusion: the power and precision of ketogenic diet science
The ketogenic diet is a profound metabolic intervention rooted in solid ketogenic diet science, originally developed for therapeutic use in epilepsy. By severely restricting carbohydrates, the body successfully transitions from burning glucose to relying on fat and the subsequent production of ketone bodies — nutritional ketosis. These ketones serve as a highly efficient “super fuel” for both the body and the brain, driving rapid short-term weight loss, improving glucose control and insulin sensitivity, and offering neuroprotective benefits.
However, the power of the ketogenic diet comes with inherent risks — including the potential for nutrient deficiencies, kidney stone formation, and long-term cardiovascular concerns, particularly if the diet is high in poor-quality saturated fats. Due to its highly restrictive nature and complex metabolic impact, the KD is not a casual or universally sustainable long-term solution for the general population.
It is imperative that anyone considering a ketogenic diet consults with a specialized healthcare professional to ensure the diet is medically appropriate for their individual health status and that proper nutritional and metabolic monitoring is maintained throughout.
Related resources & tools
The foundational explainer on ketosis — what triggers it, how long it takes, and how to know you’re in it.
The most important safety distinction in ketogenic nutrition — explained clearly and without confusion.
Translate the science into personalized fat, protein, and carb targets for your specific body and goals.
Compare testing methods for accuracy and learn which meter provides the most reliable BHB readings.
Everything you need to navigate the adaptation phase — with specific electrolyte dosing protocols.
Which ketogenic protocol fits your lifestyle and goals? The full breakdown of every major variant.
Explore more keto science & diet resources
For additional information, clinical studies, and comprehensive guides on ketogenic diet science, check out these authoritative external resources:
